The researchers created 64 mutant strains missing different combinations of the worm’s six serotonin receptors to determine their functions. Three receptors caused serotonin release slowly, while the other three regulated the major receptors.
Serotonin release patterns affected distinct receptors. The scientists studied serotonin’s circuit-level effects by fluorescently labeling each receptor gene in each brain cell. The study shows the challenges and potential of serotonergic-targeted psychiatric medications.
Psychiatric medications target serotonin, a brain neurotransmitter that affects mood and behavior. Scientists need to understand how the chemical impacts brain cells and circuits in health and sickness to develop better treatments.
Researchers at MIT’s Picower Institute for Learning and Memory used a basic animal model to explore how serotonin impacts behavior from molecules to the animal’s brain.
“There have been major challenges in rationally developing psychiatric drugs that target the serotonergic system,” said Steve Flavell, associate professor in The Picower Institute and MIT’s Department of Brain and Cognitive Sciences and senior author of the Cell paper. Complexity abounds.
There are several kinds of serotonergic neurons with extensive brain projections, and serotonin operates through multiple receptors, which are typically activated together to modify neuronal circuits.”
Scientists encounter these similar problems in C. elegans, but to a lesser extent. C. elegans possesses 302 neurons and six serotonin receptors, compared to 14 in humans. C. elegans neurons and connections have been mapped, and its cells can be genetically manipulated.
Finally, Flavell’s team can track and visualize worm brain neuronal activity concurrently using imaging technology. For these reasons, the group was able to perform a breakthrough study showing how serotonin’s widespread biochemical action influences brain-wide activity and behavior.
“These results provide a global view of how serotonin acts on a diverse set of receptors distributed across a connectome to modulate brain-wide activity and behavior,” the researchers wrote in Cell.
Ugur Dag, Di Kang, and Yale MD-PhD student Ijeoma Nwabudike are the study’s co-lead authors.
Savoring slowing
In 2013, Flavell revealed in Cell that C. elegans slows down when it reaches food by using serotonin from an NSM neuron. The new study employed MIT’s numerous new tools to extensively explore serotonin’s impacts.
First, they identified the worm’s six serotonin receptors’ functions. They developed 64 mutant strains to cover all receptor knockout combinations. One strain had one receptor taken off, another had all but one, and another had three. The scientists induced NSM neuron serotonin release to halt each worm.
Two significant discoveries emerged from data analysis: Three receptors largely slowed behavior. The other three receptors “interacted” with slowing receptors and altered their action. Flavell said psychiatric medicines that target serotonin receptors may be directly affected by these complicated behavior-controlling relationships.
Serotonin’s activities were also illuminated. One was that living animals’ receptors release serotonin differently. The SER-4 receptor only reacted to abrupt NSM neuron serotonin release. However, NSM-induced “tonic” serotonin release activated the MOD-1 receptor. This shows that living animals use various serotonin receptors at different times.
Brain-wide mapping
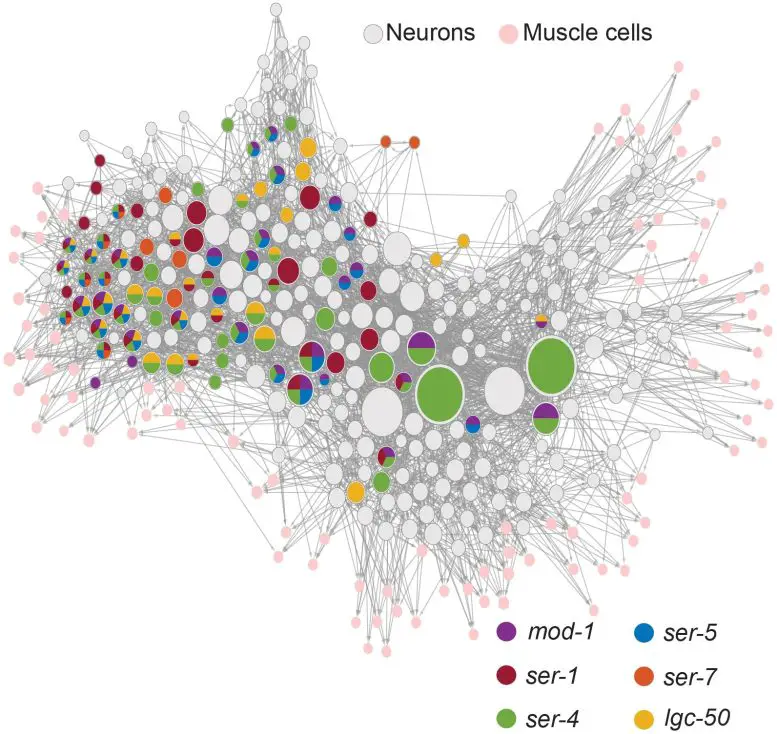
After identifying the roles of serotonin receptors in C. elegans behavior, the researchers employed imaging to investigate how serotonin affected circuits. For example, they fluorescently tagged each receptor gene in each neuron across the brain to view all the cells that produced each receptor, creating a brain-wide map of C. elegans serotonin receptors. Some worm neurons express five kinds of serotonin receptors.
Finally, the scientists tracked all neuron activity (calcium fluctuations) and behaviors to see how the serotonergic neuron NSM influenced other cells while worms freely explored their environment. Serotonin released half the worm’s brain neurons. Since they knew whose neurons they were recording from, the researchers wondered if knowing which serotonin receptors each cell expressed might predict how they responded to serotonin. Knowing each neuron’s receptors and input neurons predicted how serotonin affected it.
“We performed brain-wide calcium imaging in freely-moving animals with knowledge of cellular identity during serotonin release, providing, for the first time, a view of how serotonin release is associated with changes in activity across the defined cell types of an animal’s brain,” the researchers concluded.
Flavell said these data illuminate medication developers’ challenges and prospects. The study found that targeting one serotonin receptor may depend on other receptors or cell types. The study shows how serotonin receptors influence neuronal circuit function.

